 
If you experience mild to moderate side effects after the first dose, that doesn’t mean you should skip the second dose. It’s important to recognize that some side effects from the Pfizer-BioNTech COVID-19 vaccine are common and normal. We need to continue to collect data to make sure it stays that way,” he said. “Of course, we have to continue to monitor going forward. Matthew Heinz, a hospitalist based in Tucson, Arizona, told Healthline. “Based on the large number of folks that have been closely monitored during the study, the patient safety profile is excellent in terms of side effects,” Dr. So far, research has also found that the vaccine has a good safety profile. The available data suggests that after two doses, the vaccine is 95 percent effective at preventing COVID-19. Based on evidence from ongoing clinical trials, the FDA found that the known and potential benefits of the vaccine outweigh the known and potential risks. The FDA issued this authorization after reviewing the available efficacy and safety data on the new vaccine. Under this EUA, Pfizer-BioNTech’s new COVID-19 vaccine can now be distributed in the United States. Other commonly reported side effects of the Pfizer-BioNTech COVID-19 vaccine include fatigue, headache, and muscle pain.Įarlier this month, the Food and Drug Administration issued its first emergency use authorization (EUA) for a vaccine against COVID-19 in people aged 16 years and older. 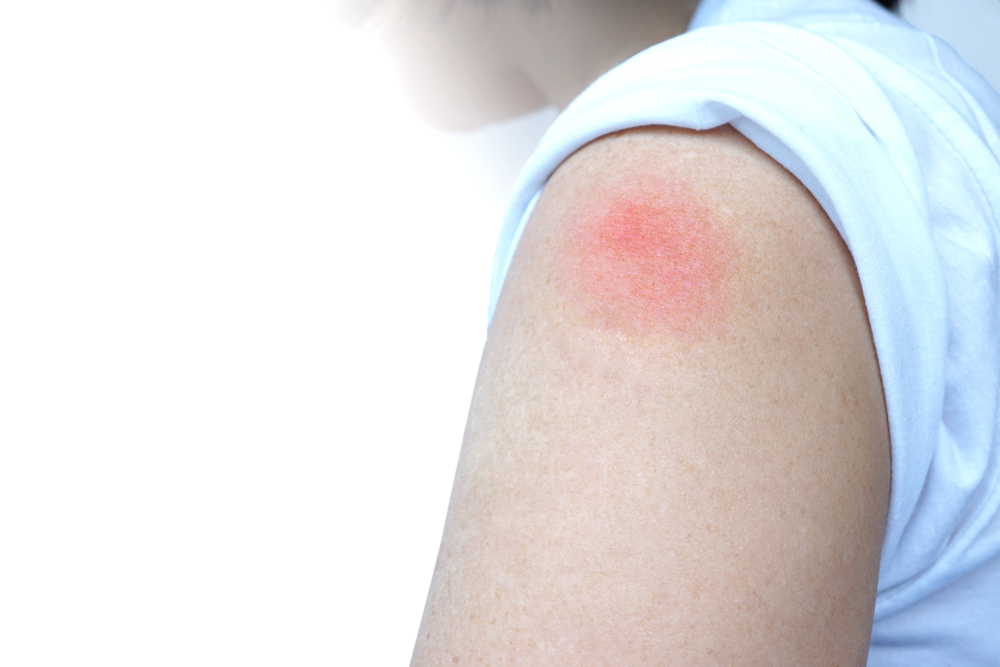
The most commonly reported side effect from the vaccine is injection site reaction.To date, safety data on the vaccine has been collected from 37,586 participants enrolled in an ongoing phase 3 clinical trial.Share on Pinterest Healthcare workers across the nation are getting the COVID-19 vaccine. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
